|
7/21/2023 0 Comments Capa report
Containment is basically the immediate process or measures taken to prevent further use or distribution of non-conforming material. In the event that a non-conformance is identified, the issue should be “contained” and it warrants a root cause analysis. This article breaks down the concepts of Correction, Corrective and Preventive Action and explains how they tie together with the CER and PMS.ĬAPA Management: Containment and Root Cause Analysis Even experienced and knowledgeable quality experts can slip among these during the stress induced by a recently discovered nonconformance. However, there is a general lack of understanding between the different types of measures taken to address non-conformance such as corrections, CAPA and containment. 
While the CER must detail preventive measures taken to ensure the safety and efficacy of the device under evaluation, PMS should include CAPA measures through Vigilance and Post market clinical follow up(PMCF), surveys etc. Preventive actions should also be simultaneously integrated in CAPA management for best outcome.Ĭorrective and preventive actions (CAPA) should seamlessly integrate with both the Clinical Evaluation Report (CER) and Post Market Surveillance (PMS) to ensure the greatest degree of safety and efficacy for both the patient and the user. 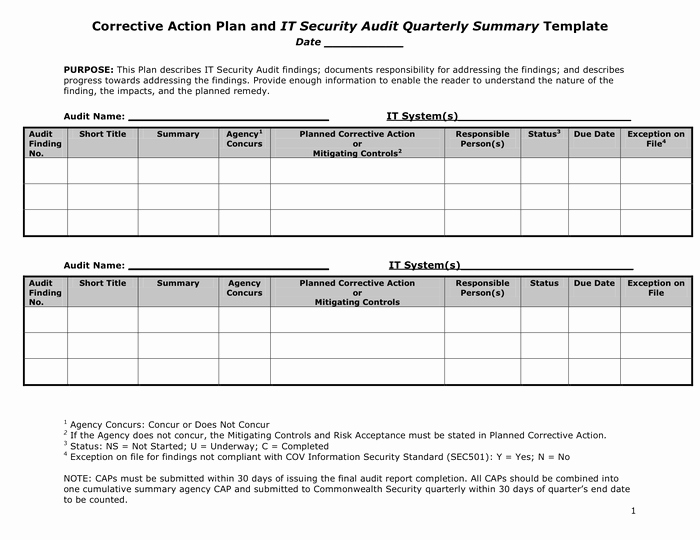
The EU MDR emphasizes field safety corrective actions to correct non-conformances of the distributed product, but since these principles are used throughout the quality system, they are not limited to the distributed product alone. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
